|
Specify the documents associated with the procedure.Ensure compliance to all ISO 13485 requirements.Include specific actions, controls, and interactions.Identifying the right teams for individual work and schedules.The team explains the individual roles and responsibilities of stakeholder teams.Ensuring that there is consistency in terminology.Removing all doubt related to terminologies and definitions relevant to the standard procedures.Identifying the procedures for timely implementation and covering the standard operating procedure (SOP).Determining the scope of the program for quality management.Explaining all the critical requirements of the ISO 13485 standardĭetermining the scope of the certification.Stating the objectives and goal of getting the ISO 13485 standard.The following is the basic model that you can apply in your company. For that, a stringent plan is to be implemented. Your consumers would stop shifting to other brands when your medical devices exceed their satisfaction curve. A constant supply of quality products allows your brand to stand against of its competitors. Besides, a stable profit margin and opportunity for market exposure, it is necessary to maintaining this certification. With the help of the accreditation, a company can exhibit its dedication and commitment to the target audience. The ISO standard is mandatory as the market competition is high and there is space and prospect for entrepreneurs and small companies. Developing Standard Operating Procedures (SOPs) is a critical component of implementing and maintaining the ISO 13485 compliant QMS. The ISO 13485 is an international standard that specifies requirements for a quality management system (QMS) for medical devices. The ISO 13485 standard operating procedure is comprised of a set of activities, which help a company’s management team strictly follow the rules for quality assurance. From design to delivery of quality medical devices, a company needs a properly streamlined process that aligns to all company objectives and internationally established quality standards. The standard fundamentally provides a set of guidelines for operating systems. However, the answer is wide and more complicated. Why is the ISO 13485 a necessary standard to follow for an operating business in the medical device manufacturing and supply industry? If you run an online search, you will find countless answers, referring to the fact that the ISO 13485 is included inside any quality management standard group, crafted specifically for the medical device sector. BSI, together with its Group Companies, also offers a broad portfolio of business solutions other than NSB activity that help businesses worldwide to improve results through Standards-based best practice (such as certification, self-assessment tool, software, product testing, information products and training).ISO 13485 Standard Operating Procedure: Guide 101 The British Standards Institution (BSI, a company incorporated by Royal Charter), performs the National Standards Body (NSB) activity in the UK. Likewise, we do not offer consultancy to clients when they also seek certification to the same management system. It means decisions are made free from any engagements of influences which could affect the objectivity of decision making.Īs an accredited certification body, BSI Assurance cannot offer certification to clients where they have also received consultancy from another part of the BSI Group for the same management system. Impartiality means acting fairly and equitably in its dealings with people and in all business operations. Impartiality is the governing principle of how BSI provides its services. Delegates must pass the examination to be awarded the qualification. 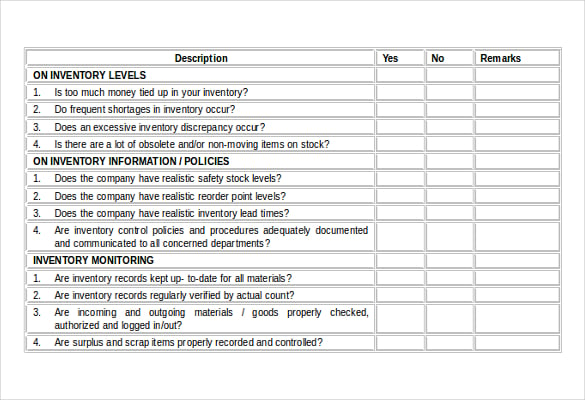
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
